Chemical Structure : NN501
CAS No.:
NN501
(NN0489-0501)
Catalog No.: PC-26157Not For Human Use, Lab Use Only.
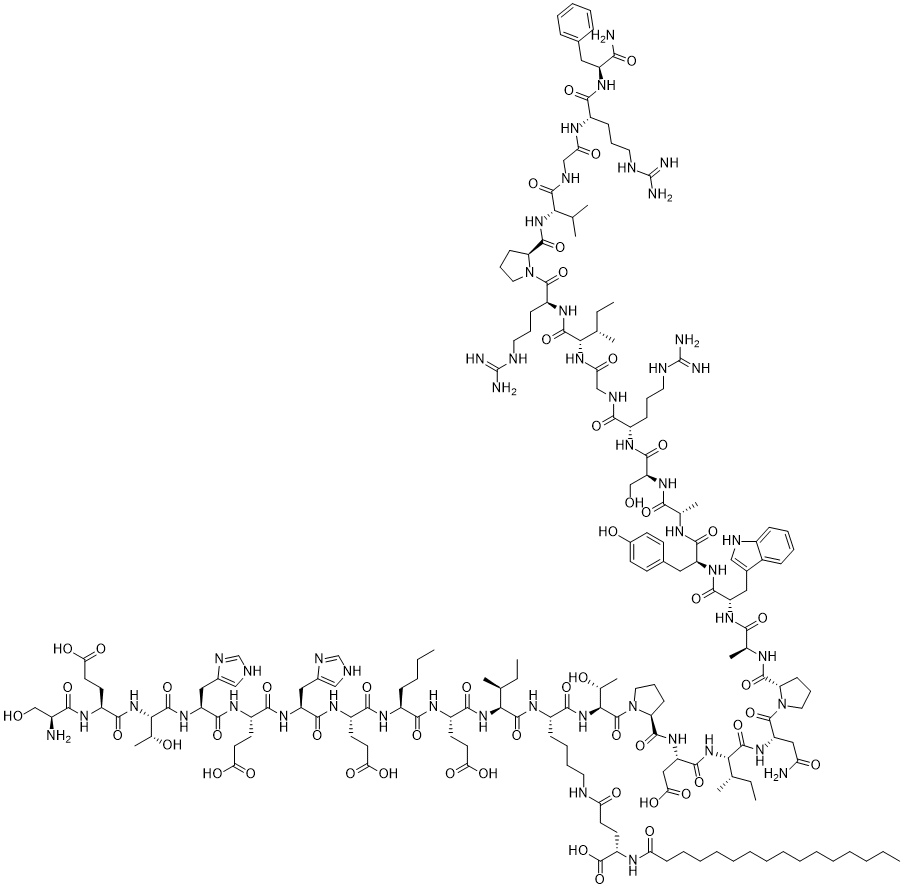
NN501 (NN0489-0501) is a potent, dual-acting pepetide agonist of GPR10 and neuropeptide FF receptor 2 (NPFFR2) with EC50 of 0.7 nM and 9.1 nM for hGPR10 and hNPFFR2 respecitvely.
Bulk size, bulk discount!
E-mail: sales@probechem.com
Tech Support: tech@probechem.com
Purity & Documentation Purity:
>98% (HPLC)
Biological Activity
NN501 (NN0489-0501) is a potent, dual-acting pepetide agonist of GPR10 and neuropeptide FF receptor 2 (NPFFR2) with EC50 of 0.7 nM and 9.1 nM for hGPR10 and hNPFFR2 respecitvely.
NN501 shows much weaker activity against hNPFFR1 (EC50=148 nM).
NN501 reduces body weight and results in a slow rate of regain after discontinued treatment.
NN501 (500-4,000 nmol/kg, subcutaneous (s.c.) dosing daily) reduction in body weight associated with a minor effect on food intake.
NN501 does not produce any obvious gastrointestinal adverse reactions or loss of lean mass.
NN501 increases energy expenditure on treatment and has a sustained effect on fatty-acid oxidation.
Physicochemical Properties
|
M.Wt
|
3973.56
|
|
Formula
|
C182H283N49O51
|
|
Appearance
|
Solid
|
|
CAS No.
|
|
|
Storage
|
- Solide Powder
-
-20°C 12 Months; 4°C 6 Months
|
- In Solvent
-
-80°C 6 Months; -20°C 6 Months
|
|
Shipping
|
|
|
Solubility
|
10 mM in DMSO
|
|
Chemical Name/SMILES
|
Ser-Glu-Thr-His-Glu-His-Glu-Ala-Glu-Ile-Lys-Thr-Pro-Asp-Ile-Asn-Pro-Ala-Trp-Tyr-Ala-Ser-Arg-Gly-Ile-Arg-Pro-Val-Gly-Arg-Phe ((Arg with palmitoylation) O=C(N[C@@H](CCC(O)=O)C(N[C@@H]([C@H](O)C)C(N[C@@H](CC1=CNC=N1)C(N[C@@H](CCC(O)=O)C(N[C@@H](CC2=CNC=N2)C(N[C@@H](CCC(O)=O)C(N[C@@H](CCCC)C(N[C@@H](CCC(O)=O)C(N[C@@H]([C@@H](C)CC)C(N[C@@H](CCCCNC(CC[C@@H](C(O)=O)NC(CCCCCCCCCCCCCCC)=O)=O)C(N[C@@H]([C@H](O)C)C(N3[C@@H](CCC3)C(N[C@@H](CC(O)=O)C(N[C@@H]([C@@H](C)CC)C(N[C@@H](CC(N)=O)C(N4[C@@H](CCC4)C(
|
References
1. Feetham CH, et al. Cell Metab. 2026 Jan 6;38(1):100-114.e6.
Contact Us
sales@probechem.com